The world's first galvanic cell was developed by Luigi Galvani. Read about its history. in this articleEssentially, it's a temporary source of electric current generated by a chemical reaction. The flow of electrons is generated by the interaction between two dissimilar metals. As a result, chemical energy is converted into electrical energy, which can then be used in everyday life.
A concentration galvanic cell is a current source that consists of two identical metal electrodes placed in a mixture of salts of this metal in various concentrations.
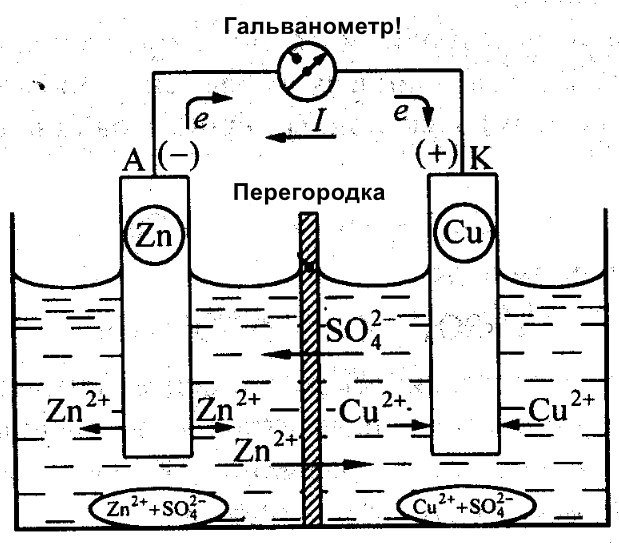
Besides Galvani, Daniel Jacobi also worked on creating an efficient battery. He slightly modified his energy source. It consists of a copper plate placed in CuSO4 and a zinc plate immersed in ZnSO4. To prevent them from interacting directly, a porous wall is placed between them. Below is a diagram of Daniel Jacobi's galvanic cell.
Zinc and copper have different reactivity, and therefore their charge will differ. As a result, the electrodes' levels are also not uniform. This allows them to move and generate an electric or galvanic current. This current begins to flow when a person or the inventor of a current-storing device connects a load. This load could be a light bulb, a radio, a computer mouse, or other electrical devices.
Schematic diagram of a galvanic cell
A circuit diagram refers to its composition and structure. It can be constructed from several chemical elements using auxiliary devices. Below, the structure of a galvanic cell will be briefly described. Read more about it in this article.!
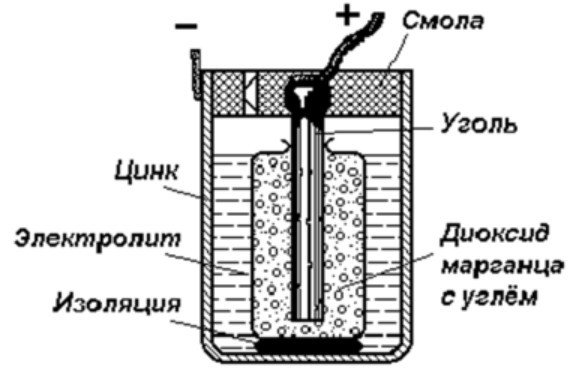
The structure of a galvanic cell
The simplest energy storage device consists of:
- Rod of coal.
- Two dissimilar metals.
- Electrolyte.
- Resin or plastic.
- Isolator.
As can be seen from this diagram, a galvanic cell consists of a negative and a positive electrode. They can be made of copper, zinc, or other metals. They are called copper-zinc cells. They are sometimes called dry batteries.
Designation of a galvanic cell in a diagram It is made in the form of two vertical lines, close to each other at a small distance. One of them will be smaller. At the edges of each such line there are signs indicating polarityThe long line is marked positive, and the short line negative. The voltage may be shown next to it. This means the circuit using the battery operates only at that voltage.
The operating principle of a galvanic cell
A galvanic cell operates through the movement of electrons from one metal contact to another. A chemical transformation occurs. Read more about the thermodynamics of a galvanic cell and the formation of galvanic electricity. Here.
Answers to frequently asked questions
| Galvanic | Explanation |
| Battery | An energy source that operates through processes occurring within a confined, miniature space. Specifically, energy is generated through a chemical reaction. |
| Voltaic cell or Voltaic pile | This is an energy element first created by a scientist named Volt. |
| Process | The interaction between chemical elements that results in the formation of an electric current. |
| Discharge | This is the completion of a chemical reaction. That is, there will be no interaction between substances. Galvanic discharge is present in Warframe. Essentially, it's a modification that's in short supply. It's used for melee weapons. PolarityV2. |
| Galvanic contact | This is the contact between the electrodes and the solution. |
| Effect | The appearance of a difference between two contacts made of two types of metal. The magnitude depends on the temperature and chemistry of the conductors. Essentially, this is Volta's first law. |
| Connection/link/chain | Connecting two or more sections of an electrical circuit to a current source. |
| Galvanic charge | Filling the battery with energy. |
Galvanization is a chemical process using electric current. The reaction reduces the amount of dissolved metal cations to such an extent that they eventually form a uniform coating on a metal electrode. As a result, the item becomes more durable, small dents disappear, and its appearance becomes more attractive.
Types of galvanic cells
There are a number of specific types of batteries.
Table of galvanic cells
| Type | Voltage | Main advantages |
| Lithium | 3 V | Big capacity, high current. |
| Salt batteries or carbon-zinc | 1.5 in | The cheapest. |
| Nickel oxyhydroxyl NiOOH | 1.6 volts | High current. High capacity. |
| Alkaline or alkaline | 1.6 V | High current. Good volume. |
This topic is covered in more detail in the article. types of batteries!
Purpose of a galvanic cell
It is designed to start electrical equipment. These may include:
- Watch.
- Remote controls.
- Flashlights.
- Medical equipment.
- Laptops.
- Toys.
- Keychains.
- Telephones.
- Laser pointers.
- Calculators.
And similar things around us.
Galvanic cell at home
You can make a simple power source yourself. To do this, you'll need the following supplies:
- Plastic cup.
- Electrolyte. This can be a saline solution, soda, or citric acid diluted in water.
- Plates of two different metals. For example, aluminum and copper.
- Wires
Manufacturing process
Take a plastic cup and pour the electrolyte into it. Don't fill it to the brim; it's best to leave it 1-2 centimeters short. Attach the wires to the metal plates. Next, place the copper and aluminum plates on the edges of the container. They should be parallel to each other. Once everything is ready, you can measure the voltage with a voltmeter.
Connect the device and touch the probes to the contacts of our power source. Hold them in place until the display shows voltage. Typically, it's 0.5-0.7 volts. These values depend on the electrolyte, or more precisely, the substance used as the electrolyte.
This is how a homemade galvanic cell is made.