Today, it's extremely difficult to imagine modern life without autonomous power sources. Specifically, batteries. These sources are used to power a huge variety of electronic devices, from children's toys to computer mice.
There are several main types of power supplies:
In this article we will take a closer look at the first and one of the most frequently used types of batteries - saline or as they are also called, carbon batteries.
Formally, the first manganese-zinc battery was developed by the Frenchman D. Leclanché in 1867. However, the pioneering American company Eveready began producing carbon-zinc batteries on a production scale to solve commercial problems. Later, the monopoly Eveready the young woman violated Duracell companyAt that time, carbon batteries began to become widespread and demand for them began to grow by leaps and bounds. Thanks to this Duracellwas able to establish large-scale production.
These batteries were called carbon or carbon-zinc batteries due to the presence of a graphite rod in the device. As history progressed and scientific progress developed, other types began to appear. types of power elements More energy-efficient and reliable. However, it's worth noting that carbon batteries remain in steady demand due to their extremely low cost.
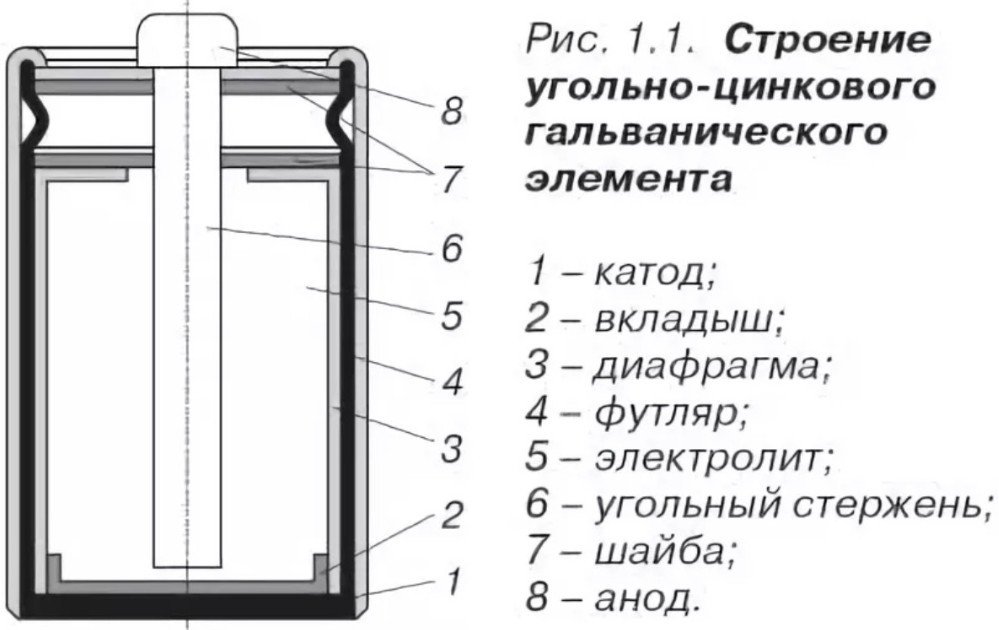
The structure of zinc-carbon batteries
The device consists of a zinc cylinder as the negative electrode, a passive carbon rod as the positive electrode, and an electrolyte (ammonium chloride). The chemical reaction produces charge carriers. When zinc-carbon batteries are not used, the charge is restored by evening out the inhomogeneities in the electrolyte composite. This increase in charge is not significant, but it does extend the life of the carbon battery.
Characteristics of zinc-carbon batteries
| Name of the indicator | Value of the indicator | Explanation |
| Battery Type | Primary or disposable | No safe recharging option |
| Battery chemical reaction type | Manganese-zinc | Used in devices with low current requirements |
| Voltage | 1.5 V | |
| Capacity | 400–1700 mAh | |
| Price category | The lowest among competitors |
Marking of carbon batteries according to international classification
Carbon batteries of foreign origin are marked with the letter R and a number characterizing the type of housing.
| Name | Marking |
| Large round elements, D | R20/LR20 |
| Small round elements, C | R14/LR14 |
| Finger elements, AA | R6/LR6 |
| Micro-finger elements, AAA | R3/LR3 |
| Elements "Krona", 9V | 6LR61 |
| Flat elements, 4.5V | 3R12/3LR12 |
As a result, we can conclude that the main and perhaps the only advantage of zinc-carbon batteries today is their low cost compared to others. types of power elementsThe disadvantages include frequent electrolyte leaks, low capacityand short storage and operating periods.
The following simple rules will help you avoid mistakes when choosing zinc-carbon batteries and extend their service life:
- If the device is not used frequently, it is better to remove the carbon batteries from the compartment.
- Carbon batteries retain their properties at temperatures ranging from 0°C to 50°C, so try to avoid purchasing batteries from open stores during the cold season.
- Carbon batteries lose up to 20% of their charge within a year of storage, so keep an eye on the manufacturing dates.
- Under no circumstances should you attempt to recharge zinc-carbon batteries. Failure to do so may result in the batteries catching fire.
- Since batteries are considered toxic waste, they must be disposed of at special facilities.
Unfortunately, such facilities are only found in fairly large cities, and the issue of proper battery disposal remains open.