Once upon a time, dry batteries were the first to be produced in large quantities. These were essentially the ancestors of scientist Leclanché. Today, these batteries are the most popular in the world. Just imagine, one company Energizermakes 6 billion in sales every year.
Dry elements have the lowest specific energy capacityOf all the batteries available on the market, this is due to their low cost. Alkaline, zinc-manganese, and zinc-chloride batteries are all dry batteries used in various electronic devices. Their names help us understand their chemical structure.
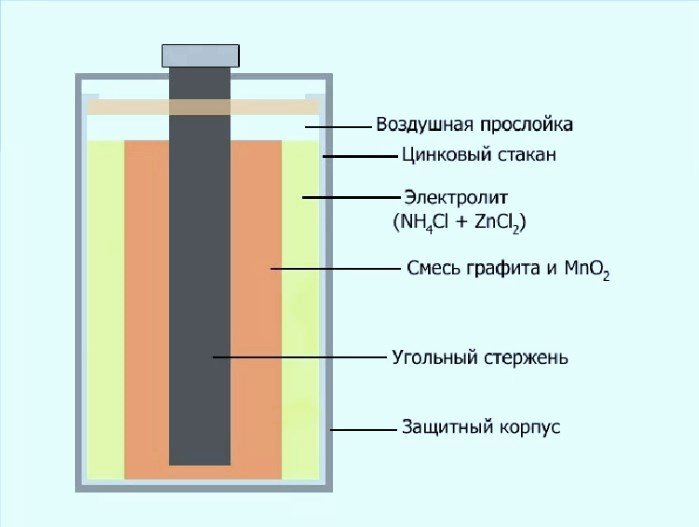
These batteries have a carbon rod along their central axis. This is the so-called cathode current collector. The cathode is essentially a complex structure consisting of a carbon-based electrode, manganese dioxide, and a special electrolyte.
The anode of a dry battery is a zinc cup. It also serves as the battery's casing. The electrolyte inside is a thickening substance. It contains manganese dioxide, ammonia, and zinc chloride.
Zinc chloride and zinc manganese batteries differ only in their internal electrolyte content. The latter contain zinc chloride and ammonia, all seasoned with water. The former are made entirely of zinc chloride. The voltage difference is very small—0.05 volts.
Can dry batteries power modern technology?
Chlorine zinc has increased capacityCompared to Leclanche batteries, this advantage disappears when applied under light load. Their attractive label reads "heavy-duty." This means they're high-power dry batteries. However, apply a heavier load, and the power of these cells drops significantly. Therefore, they simply won't power modern digital cameras. However, they'll work just fine for flashlights, TV remote controls, and other similar devices.
Alkaline batteries These are essentially carbon-zinc batteries, or dry energy storage devices, from the last century. The only difference lies in the specialized electrolytic mass. This allows for increased storage period and higher energy yield.
Why does this happen? This mixture is more alkaline than the two varieties mentioned above. The chemical composition of alkaline batteries is almost indistinguishable from that of Leclanche batteries. However, their design has distinctive differences.
Essentially, an alkaline battery is a dry cell turned inside out. Their casing isn't the anode, but rather a simple protective shell. The anode is a thick mixture of zinc powder and electrolyte. The electrolyte is an aqueous solution of potassium hydroxide.
What does the cathode consist of? Its core is manganese dioxide and carbon dioxide. It envelops the electrolyte and the anode. Using specialized technologies, scientists have learned to separate it with a layer of non-woven material. Polyester is usually used for this purpose.
How long do dry batteries last?
The device's load affects its performance. In low-demand gadgets, alkaline dry batteries can last five times longer than standard zinc-carbon batteries.
This is most noticeable when short periods of heavy use are interrupted by long periods of non-use.
If you're wondering whether a dry battery is a rechargeable battery or a battery, they're definitely batteries. Therefore, they should never be recharged. All processes occurring within them are irreversible. If you place such a power source inside, it will heat up and eventually either explode or fail completely.