The widespread use of small household appliances by consumers entails the need for power sources, or single-supply circuits galvanic cells, commonly known as batteries. They first appeared in 1800, thanks to the Italian physicist Alessandro Volta. They vary in size and shape, voltage, capacity, and battery type. Alkaline and saline batteries are widely used in the small appliance and electronics market.
What are saline batteries?
Salt batteries — sources of electric current generated in a simple device through a chemical reaction. They are sometimes called carbon-zinc batteries or carbon batteries. This type of cell is considered the cheapest, but has a low energy density, making it well-suited for low-power devices. In devices with high power consumption or with characteristic surges in load current and temperature, they do not last long and are therefore not used.
Designation of salt batteries
The designation of saline batteries is accepted according to several standards:
- American classification system.
- International system (IEC).
- Interstate standard (GOST).
| American classification system | International Classification IEC | GOST | Size, mm | Capacity, mAh |
| A | R23 | 17x50 | ||
| AA | R6 | 316 | 14.5x50.5 | 1100 |
| AAA | R03 | 286 | 10.5x44.5 | 540 |
| C | R14 | 343 | 26.2x50 | 3800 |
| D | R20 | 373 | 34.2x61.5 | 8000 |
| F | R25 | 33x91 | ||
| 1/2AA | R14250 | 312 | 14.5x25 | 250 |
| R10 | R10 | 332 | 21.5x37.3 | 1800 |
For example, the marking of a saline battery R 6 means: round, cylindrical, finger, salt type element, overall dimensions 14.5x50.5, capacity1100 mAh. Standard voltage is 1.5V. The "R" designation (salt) on the battery label distinguishes saline batteries from other types of batteries (alkaline or lithium).
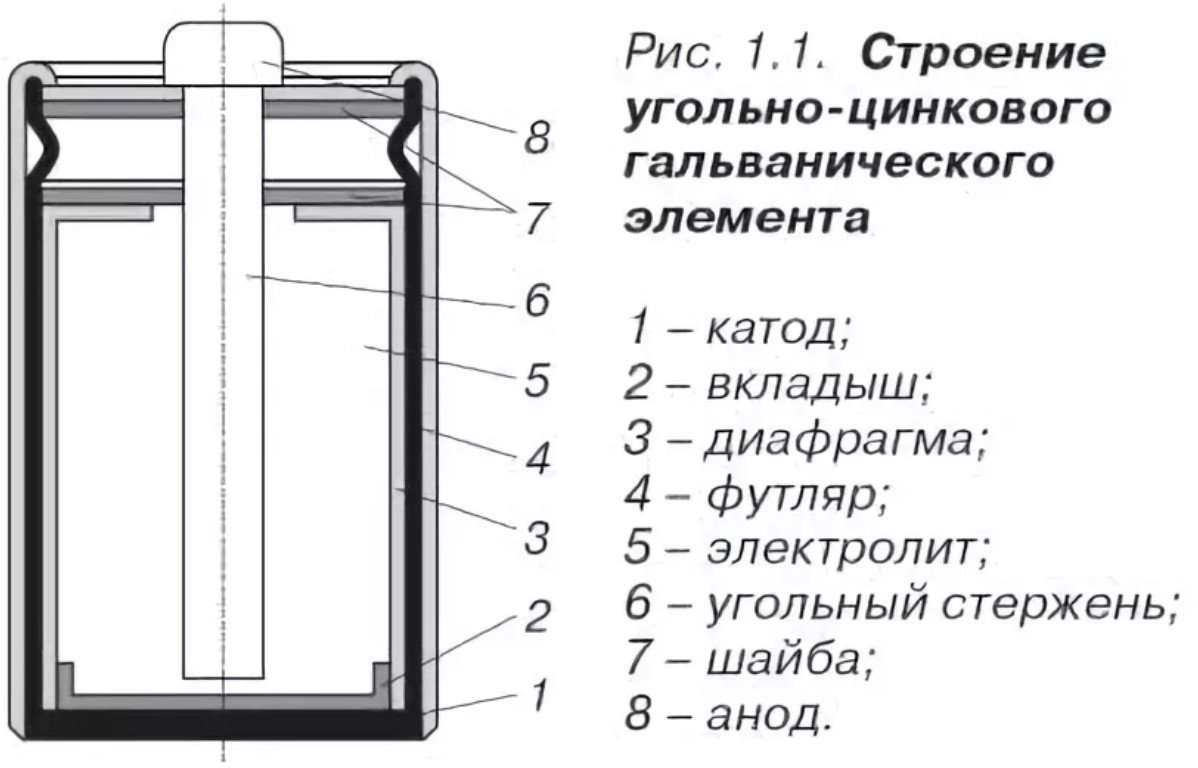
Design and composition
The design of a salt battery is quite simple and consists of:
- Cathode - the same the case of the salt battery is made made of zinc, with improved anti-corrosion properties and a high degree of purification (minus).
- The anode is an agglomerate, manufactured by pressing, impregnated with electrolyte (plus).
- Electrolyte: ammonium chloride or zinc chloride, with the addition of a thickener (starch).
- Carbon current conductor - runs through the center, treated with paraffin composition.
- Gas chamber - located at the top, designed to collect gases from the chemical reaction.
- Gasket - located at the top, acts as a sealant.
- Protective case - cardboard or tin to protect against corrosion and electrolyte leaks.
Let's consider the composition of a saline battery from a chemical point of view:
- The cathode is highly purified zinc, resistant to corrosion.
- Anode - MnO mixture2, graphite, electrolyte impregnation.
- The electrolyte is ammonium chloride, or a zinc chloride mixture with calcium chloride.
Which batteries are better: saline (1) or alkaline (2)?
The comparison can be made in the form of a table, where the advantages and disadvantages of different types of elements are clearly visible:
| Comparison parameters | 1 | 2 |
| Temperature conditions, operating conditions | Performance is reduced at low temperatures and cannot withstand current surges. | Works well at low temperatures - down to - 20O, easily withstand surges, are not afraid of a sharp increase in load current |
| Best before date | 2-3 years | 5 years |
| Application | Suitable for use in devices with low current consumption | Suitable for use in equipment with medium and high energy consumption |
| Instruments, technical devices | Alarm clocks, wall clocks, remote controls, mini flashlights, simple toys | Players, flashlights, musical toys, voice recorders, blood pressure monitors |
When choosing a power source for a device with specific operating requirements, it's important to know how to distinguish between a saline battery and an alkaline battery. While saline batteries are marked with the letter R, alkaline batteries are preceded by the letter L (LR).
Conclusions and recommendations
This type of power source is lighter and more affordable. However, the shelf life of saline batteries is short—2 to 3 years—and they can undergo partial self-discharge during storage, and during use, during a sudden current surge. Therefore, given their relatively poor performance but relatively low cost, it's easy to purchase several sets of batteries as a backup.
Not tall capacitysalt batteries (2-3 times less than other types) limits their use to simple devices with low load current.
It is easy to determine whether it is possible to charge saline batteries: the battery’s name is indicated on the body of the device intended for charging. capacityIn mAh. If this value isn't available, it's a regular battery, and despite numerous expert advice, charging it shouldn't be done. Charging it won't provide the expected long-term effect and could lead to an accident due to overheating and electrolyte leakage. If you need a more reliable, long-lasting power source, it's better to invest in an alkaline alternative or a rechargeable battery.
Interesting fact: DIY salt battery It can be made very simply. You'll need 50-kopeck coins, foil, paper, and a salt solution. It's best to soak the coins in a vinegar solution before use to remove any plaque or dirt. Assemble the device: a coin, paper soaked in the salt solution, and foil. Repeat this process several times, creating a column with the coin (positive) at one end and the foil (negative) at the other. Current is generated by the potential difference created by the electrolyte (salt solution) between the metal of the foil and the coin. This invention is called a voltaic pile, and its voltage directly depends on the number of coins used: the more coins there are, the higher the voltage obtained. However, after the experiment, the coins are no longer usable; they will have a rusty coating.