For many devices, alkaline or zinc-manganese batteries are best. galvanic cellsThese are also known as alkaline batteries. They can be found at any retailer for a reasonable price. What are they? What do they actually look like? Essentially, they're powerful batteries that undergo a vigorous chemical reaction, producing electricity.
The main element is manganese dioxide, which serves as the cathode. The anode is zinc powder. The electrolyte is potassium hydroxide, which is the alkali from which they get their name. These batteries are most often used to power toys, flashlights, radios, and other similar devices.
The use of alkali as an electrolyte was first independently proposed by two scientists, Edison and Waldemar Jungner. They then used it in nickel-cadmium batteriesEngineer Lewis Urry was the first to use this substance in conventional batteries. In 1950, he released batteries called EvereadyIn 1960, he and his partners acquired a patent for alkaline batteries.
- Characteristics
- Alkaline button batteries
- Finger-type current sources or AA
- Alkaline batteries barrels
- Pinky current sources
- Alkaline element Krona
- Main parameters
- Chemistry in alkaline batteries
- Alkaline battery structure
- Alkaline battery analogs
- Advantages of alkaline batteries
- The main disadvantages of alkaline batteries
- Which batteries are better: alkaline or saline?
- Differences between saline and alkaline batteries
- Major well-known manufacturers of alkaline batteries
- Can alkaline batteries be recharged?
- The difference between alkaline and lithium batteries
- Recommendations and advice for consumers
Characteristics
Alkaline batteries are produced in different sizes, mainly AAAA, AAA, AA, D, C, Krona - 6F22 and flat coin type.
Alkaline button batteries
They are small, flat disks with a diameter of 4.8 to 30 mm. They produce a voltage of 1.5 to 3 volts. Their casing is made entirely of metal. This allows alkyd batteries to be used in adverse weather conditions. The anode and cathode are separated by an insulator, preventing short circuits and damage. Their small size and compact size have made them widely popular. They fit easily into watches, calculators, key fobs, and other small devices.
Finger-type current sources or AA
These are the most popular. They are used where a high discharge current is required. Their voltage is 1.5 volts. They have found application in watches, toys, radios, flashlights, medical equipment, scales, computer mice, and other electronic devices.
All the details are provided in the article. AA batteries!
Alkaline batteries barrels
These include two types: D and C.
They provide electricity to a device without connecting it to a 220-volt network. They are used in tape recorders, radios, flashlights, and toys. They have increased capacityA device that consumes little energy will last a long time.
If you see an inscription on the body LR, then these are 100% alkaline batteries.
Pinky current sources
These are AAA batteries. Manufacturers produce them in the form of a cylinder with a diameter of 10.5 mm. These batteries are typically up to 44.5 mm long. They weigh approximately 14 grams. These data may vary depending on the manufacturer. Alkaline batteries may sometimes have additional protective elements added to them.
They are used in various electronic devices, such as TV remote controls, alarm clocks, flashlights, receivers, players, etc. You can tell that it is an alkaline element by the label. alkaline battery.
Learn more about these power sources read this article!
Alkaline element Krona
It has a non-standard shape and is used in measuring instruments, medical equipment, flashlights, metal detectorsThese alkaline batteries have a voltage of 9 volts and last quite a long time. The advantage of this power source is that it can provide a large current to a device while remaining compact.
Read more about the crown in this article!
Main parameters
You can immediately spot the words "alkaline battery" on the casing. This marking on alkaline batteries immediately identifies them as such.
Voltage or EMFequals 1.5 - 9 volts.
The alkaline battery capacity reaches up to 3000 mAh. This is for the largest ones.
Specific power 100-150 kW/m³.
The temperature at which current sources can operate is from – 30 to + 55 C0.
Specific energy: 65-90 W∙h/kg;
Production is carried out in countries such as Russia, the USA, China, and Japan.
Shape: cylinders, rectangles, flattened discs.
Chemistry in alkaline batteries
First, an oxidation reaction of zinc occurs at the anode. Zinc hydroxide is formed first:
Zn + 2OH− → Zn(OH)2 + 2e−
Next comes the decomposition into zinc oxide and water.
Zn(OH)2 → ZnO + H2O
As for the cathode, a reaction of reduction of manganese (IV) oxide to manganese (III) oxide occurs on it:
2MnO2 + H2O + 2e− → Mn2O3 + 2OH−
If the electrolyte is KOH, the equation will look like this:
Zn + 2KOH + 2MnO2 + 2e− → 2e− + ZnO + 2KOH + Mn2O3
When the battery dies, the alkaline electrolyte doesn't run out. This means that only a small amount is required for production. As a result, this type of power supply contains one and a half times more manganese dioxide than the same batteries. saline current elements.
Alkaline battery structure
You can read about the standard structure of a current source in this articleA more detailed description is provided there.
Current is produced when a reaction occurs between the cathode and the anode.
Alkaline battery cathode composition:
- Special binding agents – 1%.
- Acetylene black acts as graphite – about 10%.
- Manganese dioxide makes up to 85%.
- The alkaline component of potassium hydroxide is up to 35%.
The latter can also be active elements such as lithium and sodium. During the production process, the electrolyte is thickened by adding special synthetic or natural substances with a hydroxyl group (OH).
The anode is made from purified zinc. It undergoes rust-proofing procedures. Aluminum and bismuth are then mixed in.
The alkaline battery has a reverse structure when compared to saline power sources.
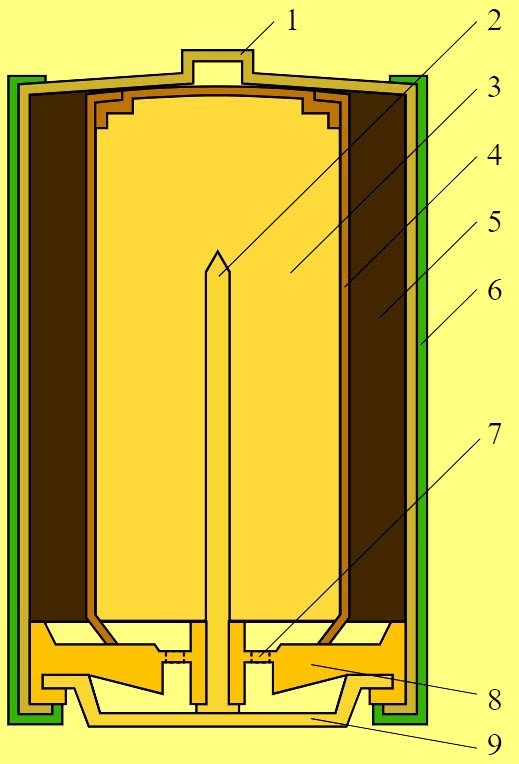
- A zinc-based paste (3) is placed inside. This metal is contained in powder form. This approach allows it to significantly increase the contact area. Therefore, capacityAlkaline batteries have a higher than saline.
- A negative potential is generated in the zinc, which is thickened with a special helium substance. This potential is applied to the rod (2), made of brass.
- Carbon from graphite, ash or soot (5) mixed with manganese dioxide is separated from zinc using a special separator device (4).
- The battery plus is a steel cup (1) coated with nickel.
- The negative pole is made in the form of a thin metal disk (9).
- The cover (6) is isolated from the main cup. This prevents a lock from occurring.
- A special gasket (8) absorbs the compressed gases. There aren't many of them, about 20%.
- A membrane (7) and a gas-absorbing chamber are installed in an alkaline battery. When pressure increases, the gases break through the membrane, causing it to rupture. At this point, the electrolyte is released.
Thus, the composition of an alkaline battery looks as described above.
Alkaline battery analogs
These power sources can be replaced with any batteries of a suitable size and voltage. The main thing is that the voltage matches, and then you can come up with something else. For example, lithium batteries can be used as an alternative. saline, mercury, silver power elements.
If we make a distribution by price category, it will look like this:
- Saline.
- Alkaline or alkaline batteries.
- Silver. Usually have a pill-shaped body.
- Lithium.
The first ones are the cheapest.
Advantages of alkaline batteries
Despite their low cost, these power supplies have a number of advantages:
- Easily replaceable, unlike batteries. No need to wait for the battery to fully charge.
- They're always available. Go to any store, be it a hardware store or a grocery store, and they're there. You'll find them at every kiosk.
- They last a long time. Some devices have built-in batteries that wear out after 2-3 years. Ultimately, if the device is non-detachable, it has to be thrown away. But battery-powered gadgets always have a replaceable battery.
- Some battery chargers take a very long time to charge, so if time is of the essence, you can easily replace them with alkaline batteries.
- Have low self-discharge.
- They perform well at low temperatures.
- They withstand strong discharge currents perfectly.
- Bthe battery is running lowevenly.
- Have high best before date.
- Safe.
The main disadvantages of alkaline batteries
Now let's talk about the downsides.
- Sometimes the price is high.
- Large mass.
- There is no possibility to use it a second time or later.
You can ignore the price, as the alkaline battery lasts a long time.
Which batteries are better: alkaline or saline?
Users sometimes debate which battery is better: saline or alkaline. A closer look reveals that alkaline batteries have a significantly higher rating! If you don't believe me, try a simple test. Buy two batteries with different electrolytes. Insert them into two identical devices. Turn them on and record the time. The one that lasts longer is the better battery!
Here are the differences between alkaline batteries and saline batteries:
- Capacity higher!
- Work 5 times longer!
- They can easily withstand temperatures down to -20 degrees Celsius!
- The electrolyte does not leak from the case when the element is deeply discharged!
So the advantages are obvious!
Differences between saline and alkaline batteries
The difference is that each one contains a different filling, and distinct chemical reactions occur within them.
Salt-type batteries use chloride salts, while alkaline batteries are filled with potassium hydroxide. The powdered metal allows the latter to last longer. As a result, the energy output has increased significantly—by as much as fivefold, according to some sources.
Another difference between alkaline batteries is their service life and operating conditions. They can withstand temperatures from -20 to +55 or even +70 degrees Celsius. The shelf life of some such batteries can reach up to 5 years. Rumors are circulating that rechargeable alkaline batteries have become available. Salt The same current sources last up to 2 years.
Major well-known manufacturers of alkaline batteries
In reality, there are numerous companies on the power supply market, all claiming to be the best. Below are the main ones:
- Camelion The company operates from China and produces high-quality alkaline batteries. They provide stable, long-lasting performance with a high discharge rate.
- Energizer – is an American company that produces power sources that maintain voltage at a constant level almost until the very end.
- Panasonic – a well-known company with decades of experience. It produces power sources that operate reliably in cold weather and deliver a stable voltage to the device.
- Duracell– is the most popular company on the market. Its alkaline batteries have low self-discharge and high capacity.
Besides these companies, there are many others that create high-quality power supplies.
Can alkaline batteries be recharged?
Alkaline batteries cannot be recharged. Once a battery is dead, it must be disposed of at a designated recycling center. Connecting it to a charger can cause a host of problems, from electrolyte leakage to explosions and burns.
The difference between alkaline and lithium batteries
The difference is that lithium batteries last 7 times longer. They can withstand temperatures down to -40 degrees Celsius and can be stored for up to 10 years. They have a higher voltage than alkaline batteries, with a voltage of 1.5, 3, 3.6, and 3.7 volts. They maintain a constant charge level and then lose it sharply at the very end. These batteries will be lighter. capacityseveral times more. Lithium batteriesThey can explode when recharged, exposed to water, or if the casing is damaged. There is also a difference in design.
But if you are wondering which batteries are better, alkaline or lithium, then it is definitely lithium-based cells.
Recommendations and advice for consumers
- Before purchasing, if you're unsure what type of battery you need, check the device's manual. The manufacturer usually specifies the battery type.
- It's better to use battery-powered power sources, as they last a long time. This is cost-effective.
- Take batteries with the voltage required by your device.
- Pay attention to the manufacturing date. Don't buy energy storage devices with a reserve.
- It's best to give preference to well-known brands. But this isn't necessary, as unknown companies produce products just as good as popular ones. After all, everyone wants their product to sell and be in demand.
- Don't throw used alkaline batteries outside or in the trash. To protect the environment, take them to designated collection points.
- The packaging should look fresh and not have any dents.