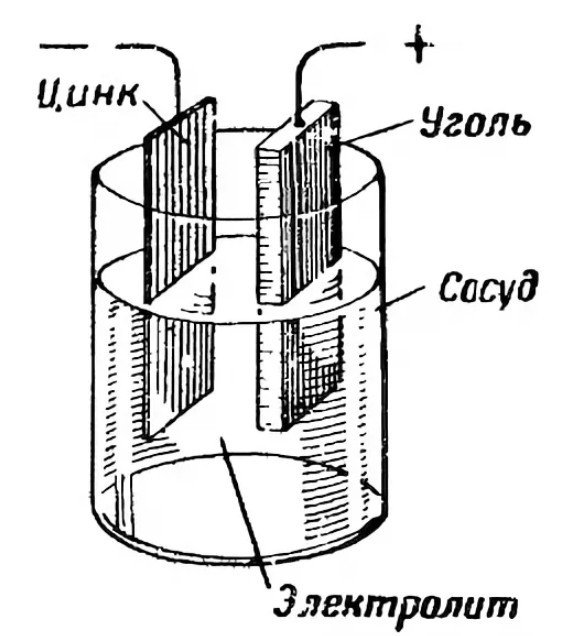
A battery operates on the principle of a simple chemical reaction, typically occurring between three elements. The resulting reaction between these substances produces an electric current. That's the gist of it.
Three key objects:
- Anode "+"
- Cathode "-"
- Electrolyte
The anode, or positive pole, serves as a source of electrons. It is usually made of zinc. The two electrodes interact with each other through an electrolyte. The electrolyte is typically a salt, ammonium chloride, or alkali. It can be dry or liquid. To thicken the substance, manufacturers add polymer compounds. Some use starch.
How a battery works
Current flows from the positive pole to the negative. This occurs when a load is connected to the battery. Simply connecting the positive and negative terminals with a wire will cause a short circuit. This can quickly drain the battery and even cause a fire.
The cathode acts as a reducing agent, acquiring electrons from the anode. In the electrolyte environment, ions move freely and facilitate efficient current generation.
What's going on chemically?
For example, in glass capacityLet's pour a sulfuric acid solution and place a zinc rod in it. Positively charged ions are present on the surface of the rod. Negative ions of the substance accumulate around the zinc object in the solution. The solution exerts attractive forces that easily detach the zinc ions. As a result, the liquid acquires a positive charge, and the zinc plate or rod acquires a negative charge. Physics tells us that potential difference equals voltage. This is what generates an electric current.
As a result, when the acid solution and the metal come into contact, an electric field is created at the interface. At this moment, chemical energy is converted into electrical energy. This is how a battery works.
After some time, the battery's life will be depleted. It all depends on where and how the power source is used. For example, if it's powering a flashlight, then with moderate use, two 1.5-volt batteries each will last for a month. However, if you insert these same batteries into an electric car, it will only run for a few hours.
As a result of all this, we can conclude that the greater the load, the faster the battery will discharge.
Read also: