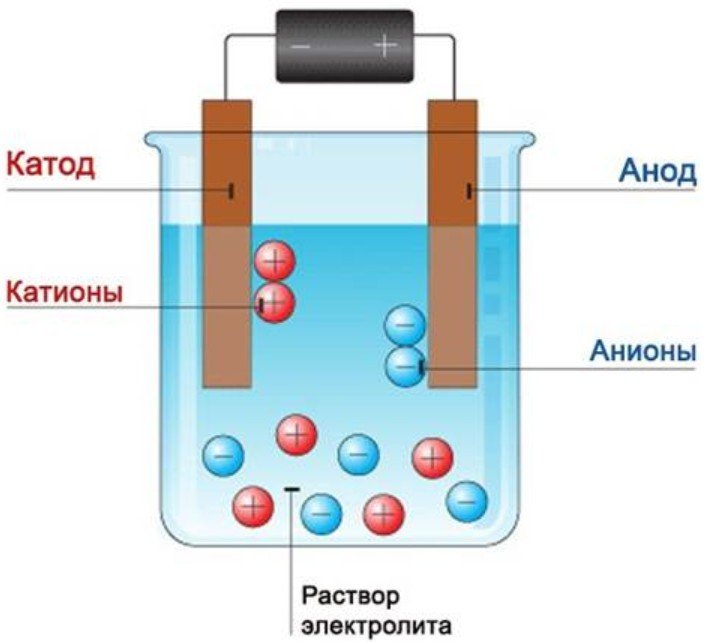
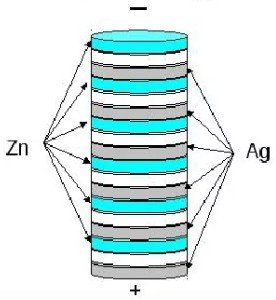
Modern people are constantly surrounded by battery-powered electrical devices. These tiny cells contain electricity. It's also present in fruits and vegetables! If you create a fruit and vegetable power source, you can charge your phone or power a lamp. Let's look at several ways to make your own battery.
Lemon battery
To make an energy source from sour fruit, you will need the following:
- 1 lemon.
- 2 wiring.
- Copper coin or wire.
- A steel nail or something else metal will help make the battery strong enough.
Read the article to learn about the production of this current source. lemon battery!
How to make a battery from glass and liquid?
To make such a power source you will need the following things:
- A glass jar or cup.
- Water.
- Wiring.
- Ammonium chloride.
- Copper plate.
- Aluminum plate or zinc.
Pay close attention to the surface area of the copper and aluminum plates. Ideally, it should be about the size of your palm. This will make the resulting battery more efficient.
Once all the equipment is assembled, solder the wires to the plates. They should be significantly higher than the can. Place them in the container so that they don't touch each other.
To obtain the correct electrolyte, mix water and ammonium chloride. Use 50 grams of powder per 0.1 H2O. Then pour this mixture into a glass or jar.
If you're not afraid, you can create an electrolytic solution using sulfuric acid. It should be 20% pure. Always pour acid into waterIf mixed up, the toxic solution will boil and spray. Don't forget to take protective equipment. Always carry gloves and goggles.
Pour the created substance to the brim into the prepared container. capacity.
If you make a battery and then replicate it several times, you can get a good device that can charge even a very energy-intensive gadget.
How to make a coin battery?
Essentially, it's a very simple battery. Some wise men call it a voltaic pile, as it resembles the first battery created by Professor Volta.
Here's what you'll need to make it:
- Copper coins. You can take 50 or 10 kopeck coins.
- Paper.
- Foil.
- Highly salted water or vinegar.
To ensure the project has a pleasing aesthetic, it's best to use identical coins. Before beginning the experiment, soak them in vinegar. This will help remove dirt and oxide deposits. Next, make coin-shaped circles out of foil and paper. There should be two fewer coins than the number of coins, as contacts are needed to attach the wires.
This coin pillar is made according to the following algorithm:
- A piece of paper soaked in vinegar or a strong salt solution sticks to the coin.
- A circle of foil is placed on top of the paper.
- After this, a coin is inserted.
- Everything is repeated until the copper coins run out.
- You should have a plus on one end and a minus on the other.
Remember that a large number of coins will increase the tension. Once this experiment is over, the coins will rust and be unusable.
When there is electrolyte between the foil and the coin, a potential difference is created and, as a result, a current is generated.
How to make a battery in a beer can?
To make this power supply you need to take:
- Salt and water.
- An aluminum can.
- Paraffin candle.
- Coal or dust from an extinguished fire.
- Graphite rod.
- Foam plastic from 1 centimeter and above.
First, cut off the top of the jar. Create a circle from a piece of foam. It should fit the bottom of the jar. You should also make a hole that's not a through hole. This hole is needed for the rod. Place the foam on the bottom and insert the graphite rod into it. Make sure it's centered exactly in the jar. Then, fill the entire empty space with charcoal.
Remember that the graphite rod should not come close to the walls of the jar, otherwise you won’t be able to make a battery.
After this procedure, all that's left is to create a salt solution. To do this, take half a liter of water and 3 tablespoons of salt. Now stir everything together and let the salt dissolve completely. Pour the resulting electrolyte into a jar and seal it with wax. Remember, the graphite rod should stick out of the jar.
Now connect the wires to the newly created terminals. The end of the protruding graphite acts as the anode, or positive terminal. The negative terminal, or cathode, will be the body of the jar. To ensure the battery generates up to 3 volts, two of these elements must be connected in series.
A battery like this could power a watch, a light bulb, or a calculator. This DIY project can even be recharged.
How to make a battery from toothpaste, potatoes, and salt?
This energy source is disposable. This element allows you to start a fire in a camping environment using a simple circuit.
Basic inventory:
- Toothpaste.
- Large potato.
- Salt.
- Copper wires without insulation at the ends.
- Small chips, toothpicks or matches sharpened to a sharp point.
Cut the potato so that the resulting surface area is as large as possible. Next, use a knife to make a hole in one half. Add salt and mix it with toothpaste. Fill the hole completely to the brim. This will be our electrolyte.
Now, take the remaining potato and make two small holes in it for the wires. They should be above our electrolyte. Push the copper wires into these holes, stripping the insulation from the ends. Now connect the two potato halves together. Finally, we've made a battery at home! Oh, and we still need to secure it with toothpicks on both ends.
Once the structure is complete, wait at least 5 minutes. Then, create a spark by short-circuiting the wires. Naturally, if you want to start a fire, the spark should be struck on something flammable.
Read also about another method making a battery from potatoes.
Of course, nothing listed above can fully replace batteries! But for fun and general development, these designs can be replicated! Fruit, vegetable, and other types of batteries are especially useful for making fires in the field!