Electrolyte is an essential part of a battery. Without this fluid, it simply won't function. In this article, we'll explain what it is, how to add it, what its specific gravity should be, and also provide some helpful tables.
- Electrolyte properties and features
- What is sulfuric acid for a battery?
- Distilled water for batteries
- Preparation of electrolyte for batteries
- Basic properties of sulfuric acid electrolyte
- Viscosity property
- Specific resistance of electrolyte
- Alkaline electrolytes
- Electrolyte shelf life
- How to check the electrolyte in a car battery?
Electrolyte properties and features
Battery electrolyte is a mixture of distilled water and sulfuric acid.
What is sulfuric acid for a battery?
In appearance, it resembles an oily substance with significant weight. It is odorless and dissolves perfectly in water. During the production of electrolyte for lead-acid batteries, heat can be observed. This effect is responsible for the dissolution of the acid.
Drivers use standard GOST 667-83 grade A acid for their batteries. High-purity acid, GOST 142b2-78, is also available in stores. It typically has the following characteristics:
- Monohydrate 92-94%.
- Density according to standard 1.830 g/cm3.
- Impurities up to 0.03665% (iron 0.012%, nitrogen oxides 0.0001%, manganese up to 0.0001%, chlorine - 0.0005%, arsenic - 0.0001%).
Distilled water for batteries
Without this water, it is simply impossible to create a high-quality electrolyte. It is prohibited to use H as a replacement.2Water from a river, a water supply, or a tap, including industrial and drinking water, is not permitted. Steam condensate is permitted. However, a chemical analysis is required before use. During this process, the iron level should be determined, and it should not exceed 0.0004%. Copper levels should also be no more than 0.005%.
Electronic distillers are used to obtain the coveted liquid in pharmacies, factories, hospitals, and laboratories. These devices are also available to the average consumer. Distilled water can be obtained using devices such as the HR-01 distiller, the Wein column still, the Pervach Econom, and others.
You should test the water produced by your distiller every 6 months. Make sure the solids levels are as follows:
- Calcium – 1.0 mg.
- Ammonia – 5 mg per liter.
- Sulfates – up to 0.5 g/l.
- Ammonium no more than 0.05 mg/l.
- Chloride not higher than 0.02 mg/l.
In addition, distilled water should be tested for nitrates, iron, and heavy metals. The resulting table should indicate whether this water can be used to create electrolytes. It must comply with GOST 6709-72.
In regular retail outlets, a 1.5-liter bottle of water can be purchased for 20-30 rubles.
Preparation of electrolyte for batteries
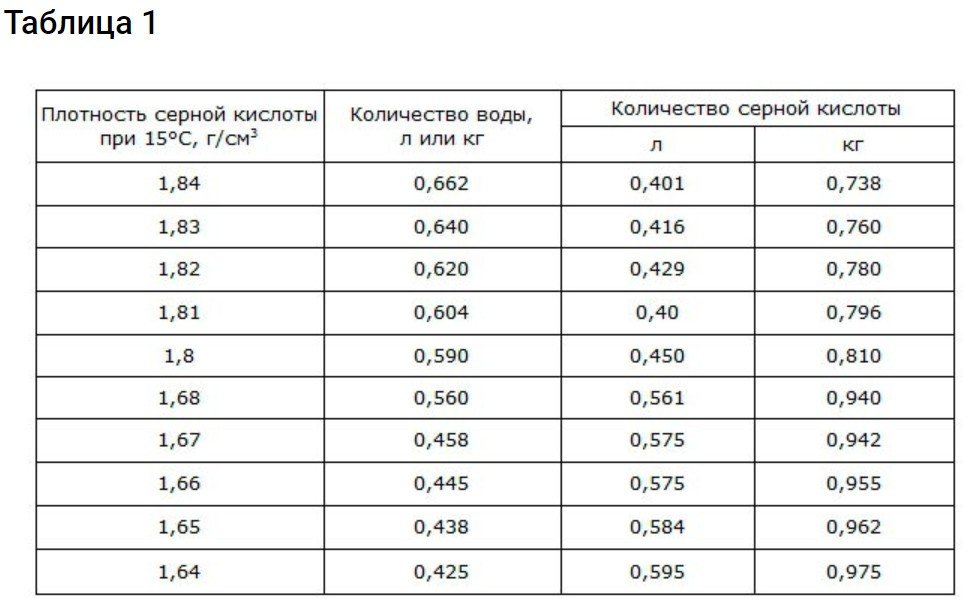
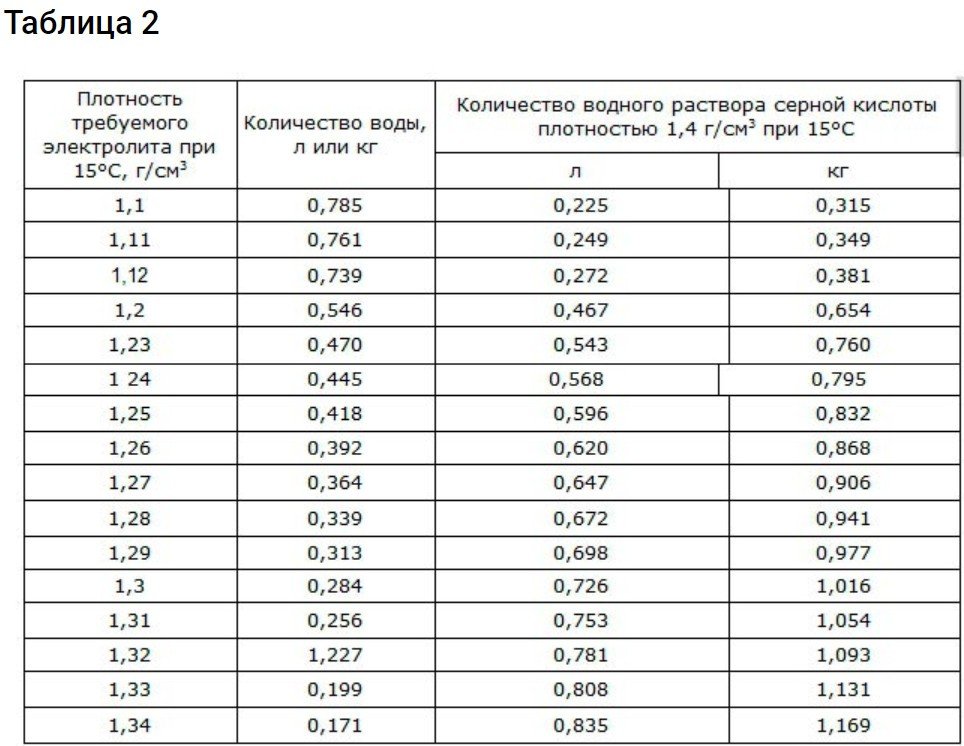
Battery electrolyte is essentially an aqueous solution of H2SO4Use water and acid with the parameters described above. For filling and topping up, use electrolyte with a density of 1.18-1.24 g/cm³. You may be using a density of 1.83 g/cm³.3 then divide the process into two approaches:
- First create an electrolyte of 1.4 g/cm3Wait until its temperature drops to 20 degrees Celsius.
- Next, make the required electrolyte from this electrolyte.
This approach will prevent the sulfuric acid from getting too hot.
The electrolyte must be prepared in a specialized container. This can be plastic, earthenware, or ebonite. Lead capacityThis will also work. But never use a glass bowl. It will crack due to the high temperatures.
The first thing you need to do is pour a certain level of distilled H2O. After this, pour in the required volume of sulfuric acid in a thin stream. Stir the mixture constantly with a glass rod. It's best if H2SO4 You will add it in portions.
Always pour acid into water. Otherwise, you'll end up with a splattering cocktail of dangerous acid droplets flying in all directions. This could cause burns to your body and damage nearby objects.
Protective equipment for work:
- Rubber boots.
- Gloves made of the same material.
- Special cloth overalls.
- Glasses for protection.
- Rubberized apron.
Below you will find special tables from which you can understand what proportions should be observed.
Data on the ratio of sulfuric acid to distilled water
To obtain a density of 1.4 g/cm3 You need to adhere to the data from the 2nd table.
To obtain a density of 1.83 g/cm3 Use the information provided below.
A hydrometer should be used to obtain density information. When measuring temperature, the thermometer should not contain any wood or metal. Measurements should be taken when the temperature drops to 20 degrees.
The temperature-density gradient corresponds to a value of 0.0007 g/cm³ per 1°C. If the temperature is above 20°C, a correction in the calculations is added to the measured density data. Let's say the actual temperature is 30°C, and ours is 20°C. Then the difference will be 10°C.0So, here's what we get: 0.0007 x 10 = 0.07 g/cm3. That is, we add an error of 0.007 g/cm3 to the density data we obtained.
If the actual temperature is 10 degrees, the difference with the temperature indicated above will be 10 C00.0007 g/cm3 is multiplied by ten and the correction is 0.007 g/cm3. In this case, the correction is subtracted from the measured density at t = 10°C.0.
It is prohibited to fill the electrolyte t more than 25 C0.
Basic properties of sulfuric acid electrolyte
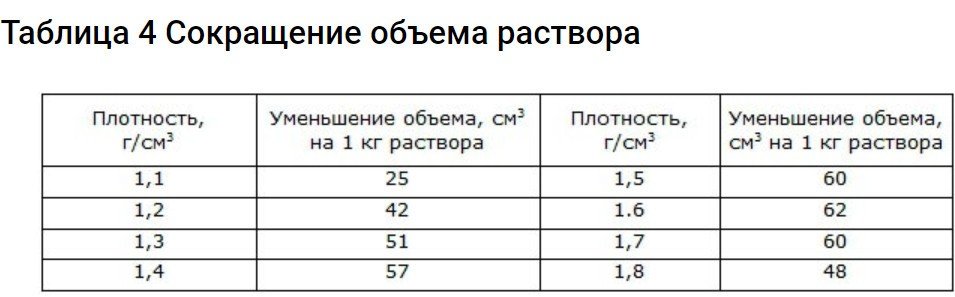
In addition to the above, another factor should be considered. If you take the same volume of acid and water, the resulting volume after mixing will be significantly smaller. Therefore, this parameter must also be taken into account when preparing the electrolyte. To do this, use the data in the table.
Viscosity property
Affects the performance of batteries with lead plates. The process in batteries is diffusional. The rate of this diffusion depends on the viscosity. This viscosity determines how quickly the electrolyte reaches the surface and pores of the electrodes during discharge.
If viscosity increases, diffusion will slow down. When temperature drops by 25 degrees, viscosity doubles. At 50°C0 its increase occurs 30 times when compared with normal t.
When viscosity increases, then battery capacitydecreases. Because of this, the performance of lead-acid batteries in cold weather decreases. This must be taken into account when installing gel batteries.
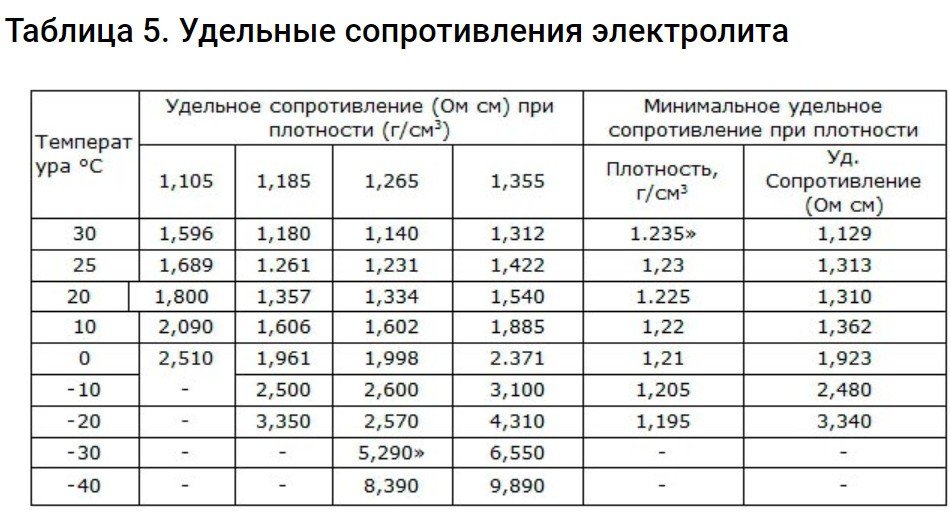
Specific resistance of electrolyte
To calculate it, you will have to resort to the formula =rS/L. It is relevant when the resistance of the electrolyte occupying capacity, limited to a length of 1 centimeter and a cross-section of 1 cm3The meaning of the letters in the formula:
- L – length.
- r – specific resistance in ohms cm.
- S – cross-section cm2.
R changes at the moment when the electrolyte temperature and concentration change. To internal resistanceIf the battery is small, you should use an electrolyte with the lowest specific R.
The parameters of such resistances are found in this table.
R increases as the temperature decreases, especially below zero degrees.
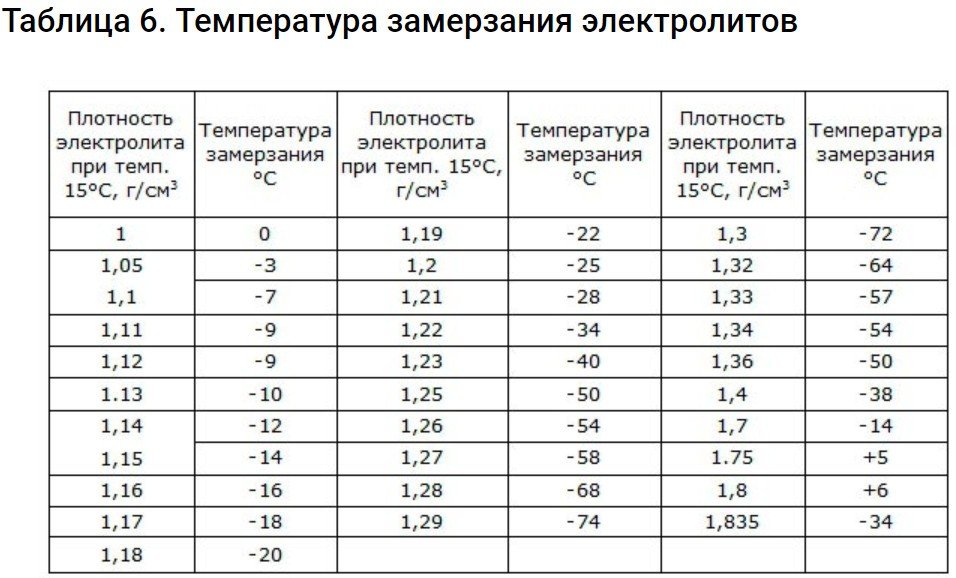
In fact, the freezing point of the electrolyte is an important indicator. As the battery discharges, its density and freezing point decrease. In cold temperatures, the volume of the liquid increases, leading to damage to the electrodes and the battery itself.
Electrolyte with a density of 1.29 g/cm3 has the lowest freezing point.
Starter batteries used in cold conditions have a density of 1.26-1.30 g/cm2This electrolyte does not freeze even in the coldest weather.
The table below shows the density levels at which freezing occurs.
Alkaline electrolytes
Lithium and potassium are used to create them; both are caustic.
Caustic potash, or KOH for short, is white, solid, and highly soluble in H2A. Heat is released when it dissolves. According to the GOST standard, this substance is produced in four grades:
- Higher.
- A
- B type.
- Jet.
The first grade contains up to 96% caustic potash. The second grade, "A," contains up to 92%. The third grade, "B," contains 88%. The fourth grade has fewer impurities than technical grade.
When creating an electrolyte from lithium and potassium, first dissolve the potassium, then add the lithium. This is done at a rate of 10-20 grams per 1 liter of electrolyte. After this, let it sit for 15-20 hours. This allows the temperature to drop and impurities to precipitate. Before leaving the solution, cover it tightly with a lid.
The resulting liquid solution should then be poured into a clean container. Next, check the specific gravity with a hydrometer. If necessary, adjust the specific gravity to the correct level. After this, add alkali and water. Alternatively, you can add a ready-made concentrated electrolyte.
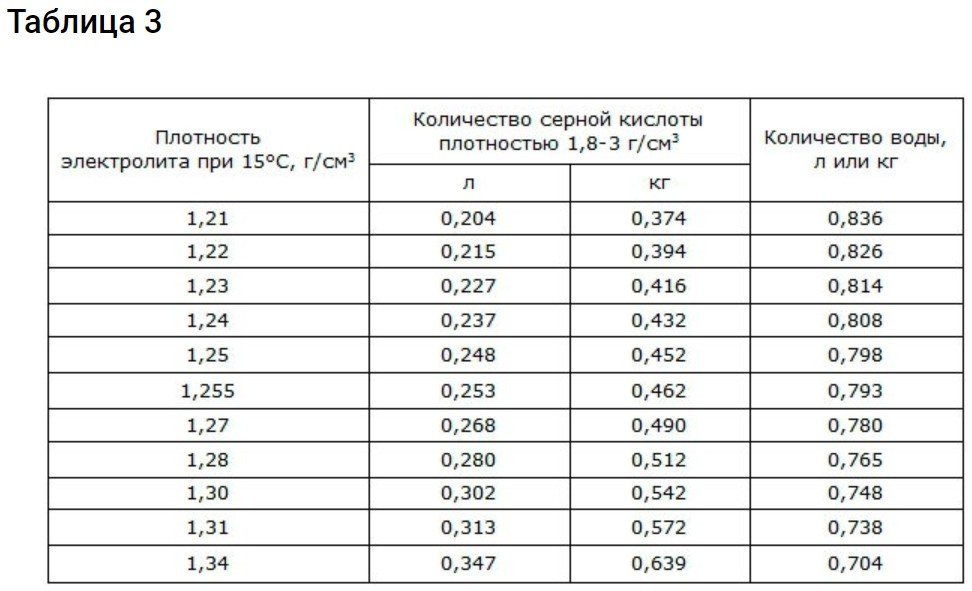
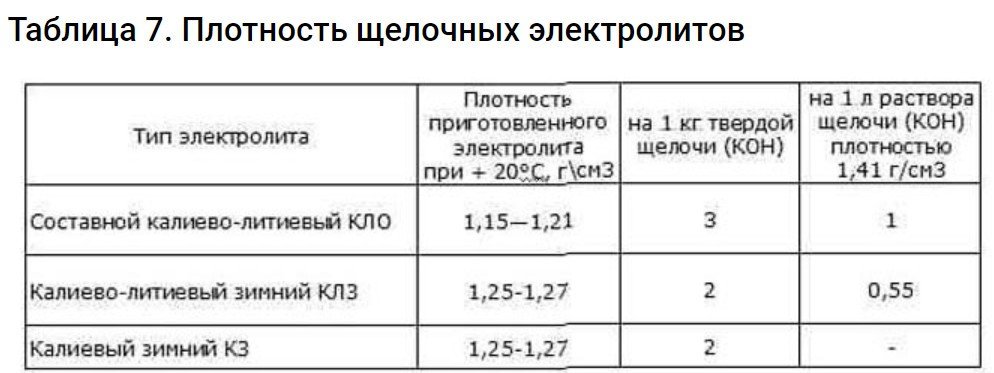
What density does your battery require? This information can be found in its technical documentation. If the documentation doesn't contain any critical recommendations, then an electrolyte of 1.19-1.21 g/cm3 can be used.3 at 15 degrees Celsius. It should also contain 10-20 g/l Li.
It's important to note that a fluid with these characteristics is best used at temperatures no lower than 20 degrees Celsius. At lower temperatures, a density of 1.25–1.27 g/cm3 is suitable.3. But without the use of caustic Li.
To revive iron-nickel and alkaline cadmium-nickel batteries, an electrolyte based on lithium and potassium with a density of 1.255-1.279 g/cm is used.3To all this, you still need to add 69 grams of caustic Li per 1 liter of electrolyte.
For ease of preparation, use the table.
Electrolyte shelf life
It can be stored indefinitely. To prevent precipitation, keep it out of direct sunlight. Also, avoid temperature fluctuations. The actual lifespan of the battery's electrolyte is unknown, as it is affected by many factors.
How to check the electrolyte in a car battery?
The check should be performed twice every 12 months: before winter and at the beginning of summer.
Tools for performing this procedure:
- Hydrometer – allows you to measure density.
- Glass tube with a diameter of 5-7 mm.
Verification algorithm:
- Remove the battery from the car.
- Unscrew all plugs.
- Place the hydrometer with the working end in one section.
- Using the bulb on the device, draw in electrolyte until the float rises and begins to float without touching the walls of the hydrometer.
- The density can be seen at the point where the rod and electrolyte touch each other.
- Write down the received information on a sheet of paper.
A similar procedure should be repeated with each battery bank.
Few people use homemade battery electrolyte these days. Simply visit any store and purchase the necessary substance for a small fee. This will save you time and protect your life!