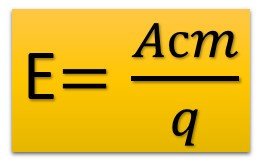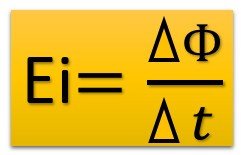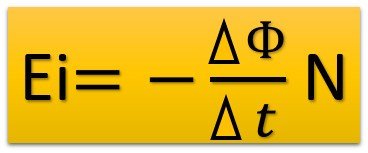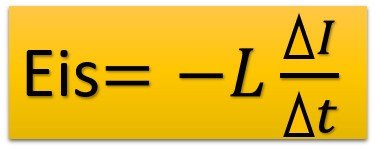In the midst of the academic year, many scientists require the EMF formula for various calculations. Experiments related to galvanic cell, also need information about electromotive force. But for beginners, understanding what it is isn't easy.
Formula for finding emf
First, let's clarify the definition. What does this abbreviation mean?
EMF, or electromotive force, is a parameter characterizing the work of any non-electrical forces operating in circuits where the current, both direct and alternating, is uniform throughout the circuit. In a coupled current-carrying circuit, EMF is equivalent to the work done by these forces to move a single positive charge along the entire circuit.
The figure below shows the emf formula.
Ast – means the work of external forces in joules.
q is the transferred charge in coulombs.
Third-party forces – these are forces that perform charge separation in the source and ultimately form a potential difference at its poles.
For this force the unit of measurement is voltIt is designated in formulas by the letter «E».
Only when there is no current in the battery, the electromotive force will be equal to the voltage at the poles.
Induction EMF:
EMF of induced in a circuit having N turns:
When moving:
Electromotive force induction in a circuit rotating in a magnetic field at a speed w:
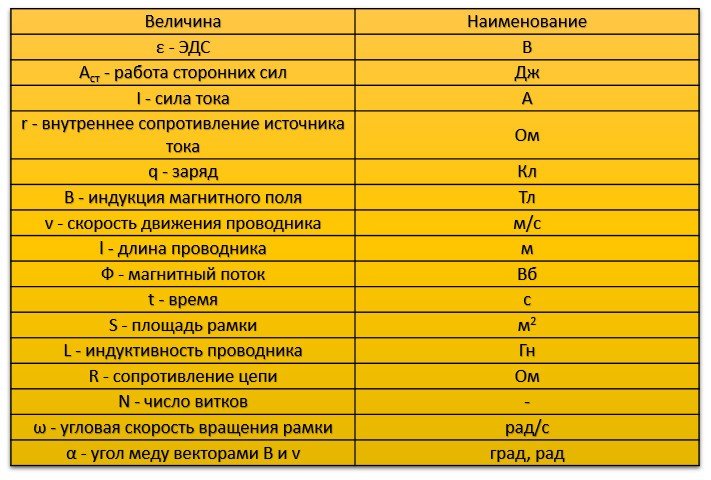
Table of values
A simple explanation of electromotive force
Let's imagine there's a water tower in our village. It's completely filled with water. Let's think of it as a regular battery. The tower is the battery!
All the water will exert a strong pressure on the bottom of our tower. But it will only be strong when the structure is completely filled with H2O.
Ultimately, the less water there is, the weaker the pressure and the less force the stream will have. When you open the faucet, you'll notice that the stream's range will decrease with each minute.
As a result of this:
- Tension is the force with which water presses down on the bottom. That is, pressure.
- Zero voltage is the bottom of the tower.
It's the same with the battery.
First, we connect the energy source to the circuit. And then, correspondingly, we close it. For example, we insert a battery into a flashlight and turn it on. Initially, we'll notice that the device glows brightly. After a while, its brightness will noticeably decrease. That is, the electromotive force has decreased (it's leaked out, compared to the water in the tower).
If we take a water tower as an example, the EMF is the pump that constantly pumps water into the tower. And the water never runs out.
EMF of a galvanic cell - formula
The electromotive force of a battery can be calculated in two ways:
- Perform a calculation using the Nernst equation. You will need to calculate the electrode potentials of each electrode included in the GE. Then calculate the EMF using the formula.
- Calculate the EMF using the Nernst formula for the total current-generating reaction occurring during the operation of the GE.
Thus, armed with these formulas, it will be easier to calculate the electromotive force of the battery.
Where are different types of EMF used?
- Piezoelectricity is used to stretch or compress materials. It is used to make quartz crystal generators and various sensors.
- Chemical is used in galvanic cellsand batteries.
- Induction occurs when a conductor crosses a magnetic field. Its properties are used in transformers, electric motors, and generators.
- Thermoelectric current is formed when contacts between dissimilar metals are heated. It has found application in refrigeration units and thermocouples.
- Photoelectricity is used to produce photocells.